High-risk parenteral products rely on high-performance CCIT solutions as even the smallest defects can pose health risks to patients. The Sepha Multi-Q HD uses advanced sensor technology that provides more sensitive, more reliable, and faster results.
Intercepting defective containers before they reach end-users or patients is important for any pharmaceutical product, but for short shelf life, highly sensitive, injectable products it is absolutely crucial. Ingress of air through micro cracks and pinholes has implications for product efficacy and safety, therefore pharmaceutical manufacturers cannot afford to be anything less than 100% confident in the integrity of their parenteral formats.
 In this post we focus on the Multi-Q HD, a bench-top Container Closure Integrity Test system that utilises vacuum decay to enable ultra-sensitive, non-destructive detection of defects in parenteral containers. We highlight the 5 key features and benefits that can help you improve quality assurance procedures, reduce manufacturing costs and enable you to meet regulatory requirements.
In this post we focus on the Multi-Q HD, a bench-top Container Closure Integrity Test system that utilises vacuum decay to enable ultra-sensitive, non-destructive detection of defects in parenteral containers. We highlight the 5 key features and benefits that can help you improve quality assurance procedures, reduce manufacturing costs and enable you to meet regulatory requirements.
1. Non-destructive Vacuum Decay Method down to 1µm
The Sepha Multi-Q HD is a non-destructive Container Closure Integrity (CCI) test system that utilises vacuum decay to detect leaks down to 1 micron in parenteral containers. Newly developed advanced sensor technology enables the Multi-Q HD to detect defects down to 1 micron (dependent on container type and content). The ultra-sensitive technology has an improved signal-to-noise ratio that can pick up the slightest rise in pressure to detect even the smallest micron holes. The sensor is located at the point of measurement, which yields a faster response and more sensitive results.
2. Deterministic & Quantitative Pass/Fail Results
Utilising the vacuum decay test method, the Multi-Q HD can identify gross, medium and micron leaks. The method provides deterministic and objective results, visualised on the result screen via a pass or fail result for each container that's being tested.
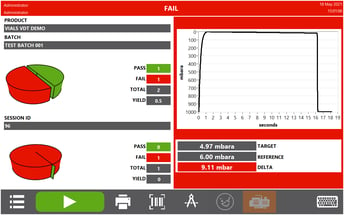
3. Utilises ASTM Standard Test Method F2338-09
ASTM F2338-09 is recognised by the FDA as the consensus standard for non-destructive detection of leaks in packages by vacuum decay method. It is applicable to test a range of non-porous flexible packages, rigid and semi-rigid packaging. The Multi-Q HD test method is a proven technology as outlined in this standard, that can be applied to different container types.
4. In line with USP 1207 and 21 CFR Part 11 Compliance
The Multi-Q HD CCIT method is in line with guidelines outlined in USP 1207 preferring deterministic and objective methods that provide reliable and repeatable results. The system also includes software developed for manufactures that need to comply with FDA 21 CFR Part 11. The software enables safe interaction between the machine, data reports and audit trails, protecting reports from being edited or deleted.
5. Ideal for Testing Vials, Ampoules, Bottles, Pre-filled Syringes & BFS
The Multi-Q HD is a multi-functional system that can work in combination with interchangeable tooling. This means the system can test an array of containers in different shapes and sizes. Depending on container type, container content and the leak rate sensitivity that needs to be achieved, our technical team will develop a method for vacuum decay testing your containers. Containers that cannot be tested using a vacuum decay test, including toxic product or oil-based liquids, can be set up for pressure decay testing.
Watch video
Interesting links


